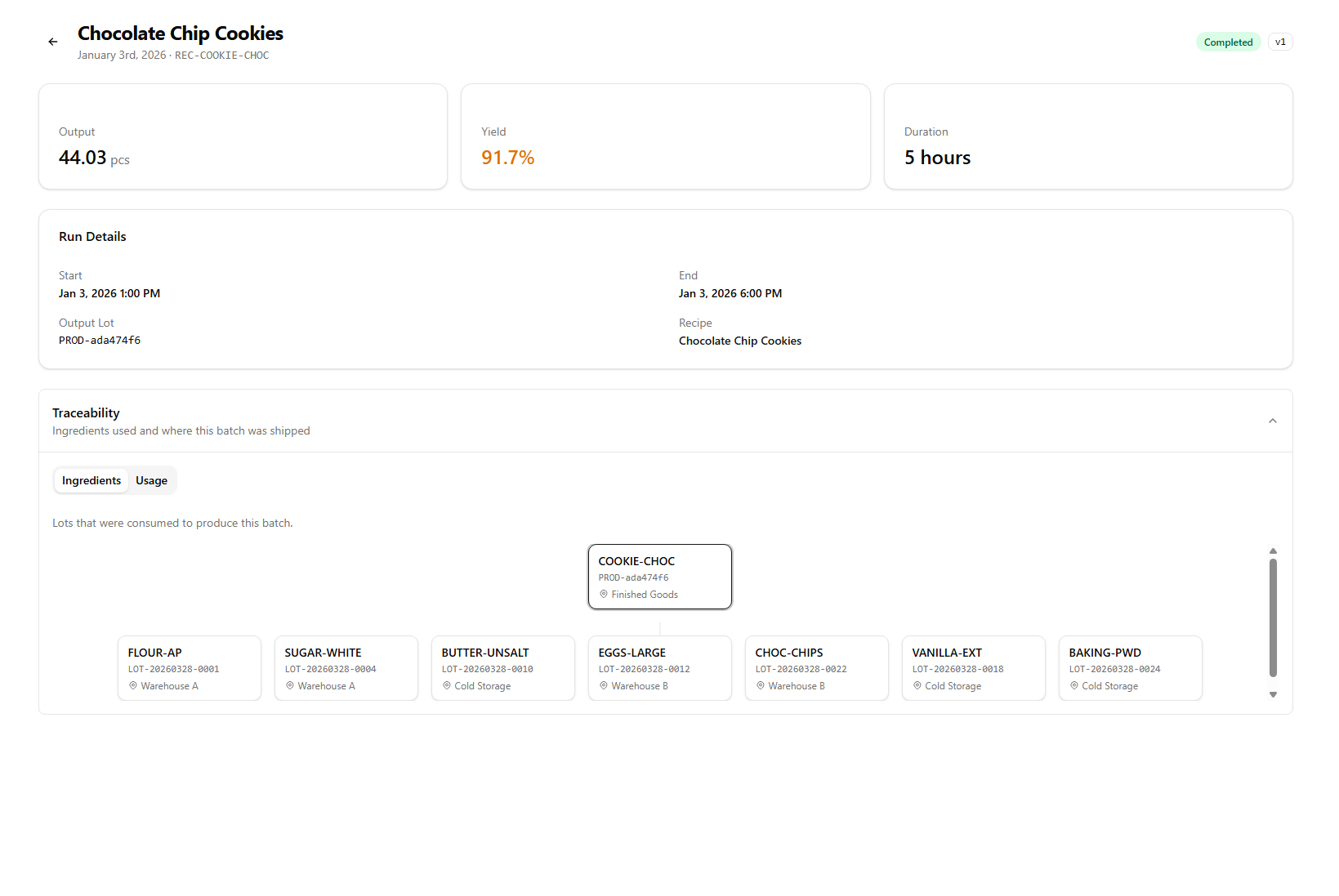
Forward and backward lot tracing
When the FDA asks "which customers received product made with lot #A-2214 of tomatoes?", you need an answer in minutes — not days. SauceControl links every supplier lot to every production run to every outbound shipment, so the full trace is one click away.
- Forward trace: raw ingredient lot → finished product → customer shipment
- Backward trace: customer complaint → batch record → supplier invoice
- Lot codes inherit through sub-assemblies — base sauce lots survive into every finished product
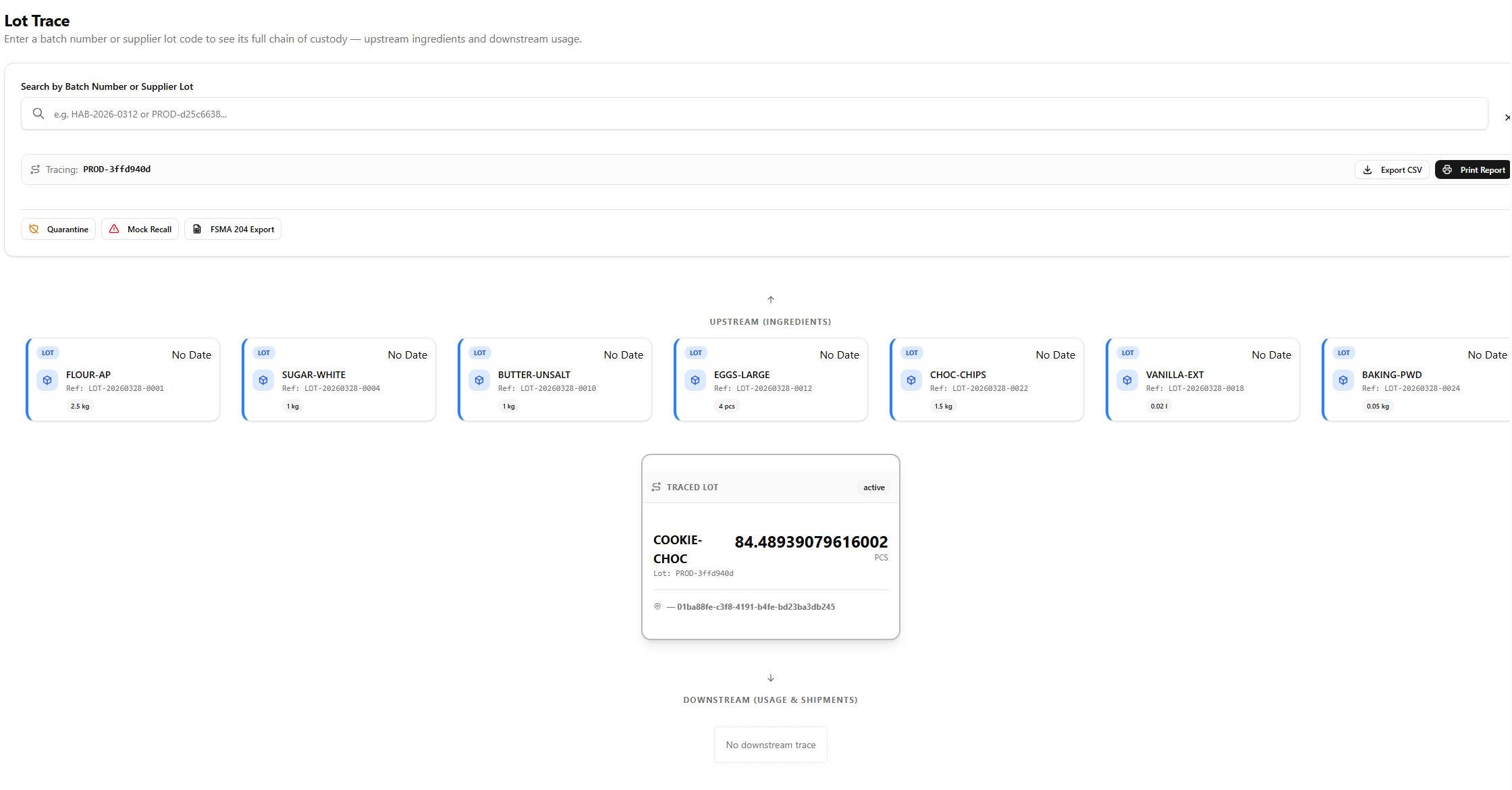
Records that build themselves
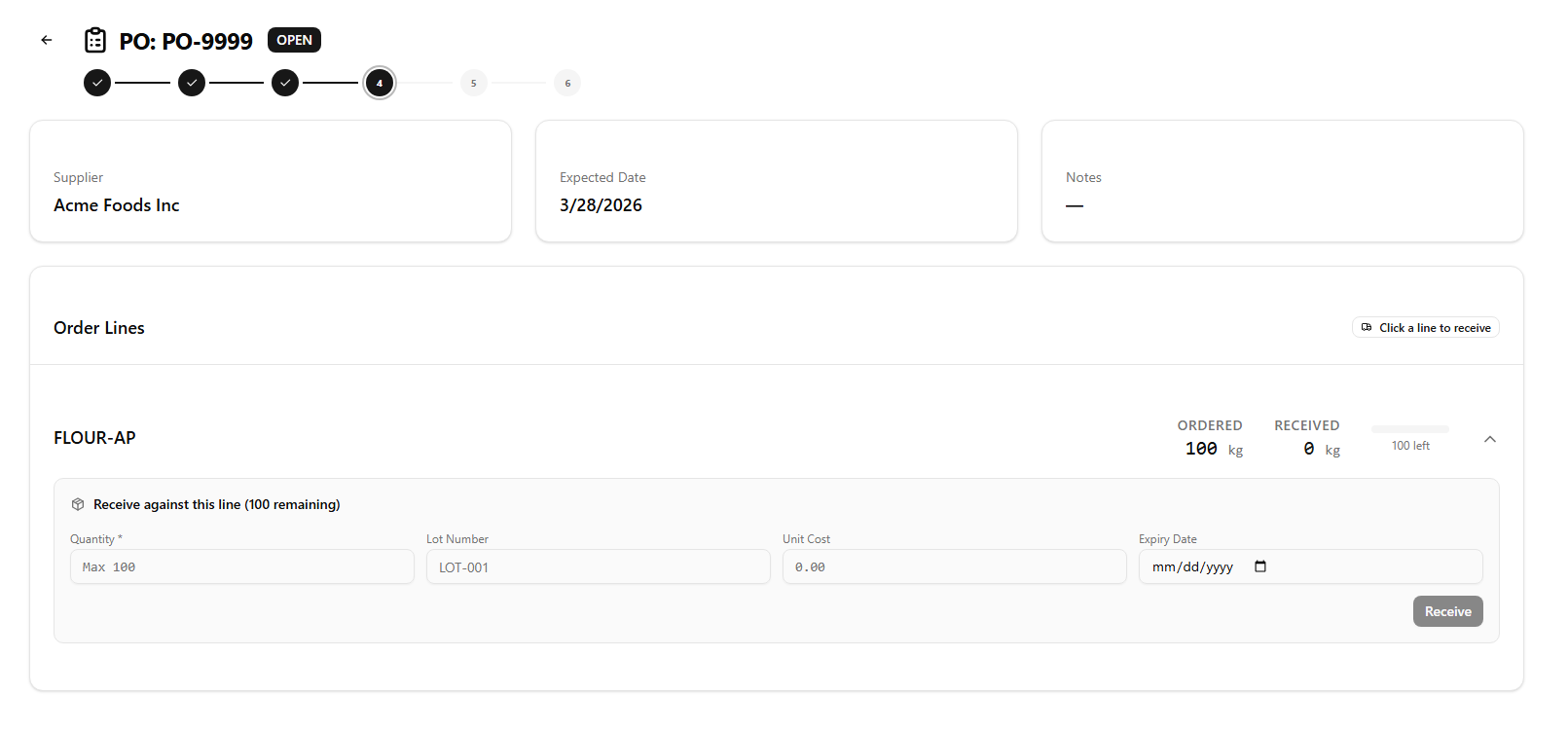
Manual log books and spreadsheets create gaps. SauceControl captures lot codes at the moment of receiving, links them to every production run that consumes them, and records them on every outbound shipment — without any extra steps from your team.
- Supplier lot codes captured at receiving — one step, permanent record
- Production runs automatically inherit lots from the ingredients consumed
- Digital audit trail replaces paper logs — always available, never lost
Mock recalls in under 2 minutes
Don't wait for a real recall to find out if your records are intact. Run a mock recall on any ingredient lot or finished product and get a complete impact report — every affected batch, every customer shipment, total quantity at risk — before an auditor ever walks through your door.
- Enter a suspect lot number — SauceControl does the rest
- Instant impact report: affected SKUs, quantities shipped, customer list
- Export FDA-format trace report — ready to hand to an inspector